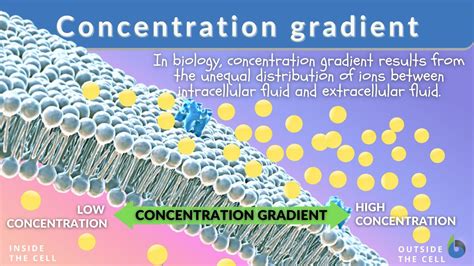
Understanding the concept of a concentration gradient is crucial for anyone looking to grasp the fundamentals of how substances move within different environments, whether in biological systems, chemical reactions, or environmental science. The concentration gradient refers to the gradient of concentration of a substance across a particular space or medium. This difference in concentration drives processes like diffusion and osmosis, which are foundational to many natural and industrial operations. In this guide, we will break down the complex concepts surrounding concentration gradients into simple, practical steps and solutions, so you can easily understand and apply these principles in real-world contexts.
Addressing the Challenges of Understanding Concentration Gradients
Many people struggle to fully grasp the concept of concentration gradients because they often see it as an abstract scientific term that doesn’t have direct, tangible implications for their daily lives. However, this notion is pivotal for various disciplines including biology, chemistry, and environmental science. The challenge lies in translating these scientific terms into a relatable, actionable form. This guide aims to bridge that gap by providing step-by-step guidance with practical, real-world examples and conversational expert advice that makes understanding concentration gradients accessible and useful.
Through this guide, you'll learn not just what a concentration gradient is, but also how to identify it in different scenarios and what actions you can take based on that understanding. This knowledge empowers you to make informed decisions in your professional and personal projects where understanding the movement and distribution of substances is essential.
Quick Reference
Quick Reference
- Immediate action item: To understand the concept of a concentration gradient, start by recognizing how substances move from areas of high concentration to areas of low concentration.
- Essential tip: A clear example is how oxygen diffuses from the lungs into the bloodstream. To grasp this, note the higher concentration of oxygen in the lungs versus the bloodstream.
- Common mistake to avoid: Confusing concentration gradient with the speed of diffusion. Remember, the gradient itself doesn’t determine the rate of diffusion but influences it.
Decoding the Basics of Concentration Gradients
At its core, a concentration gradient is a measure of how the concentration of a substance varies across a given area. Imagine placing sugar in a glass of water; initially, the sugar concentration is highest near the spoon but eventually spreads out evenly. The region where the concentration is high compared to others is known as the ‘source’ area, and where the concentration is low is the’sink’ area. This gradient drives diffusion – the natural process where particles move from a high concentration region to a low concentration region until equilibrium is reached.
To further elucidate this concept:
- Consider a scenario in a biological system like a plant's root system where water moves from soil (high concentration) to the plant cells (low concentration).
- Or think about industrial processes such as chemical reactors where a catalyst might be added to a low-concentration solution to boost a reaction rate.
Understanding this basic principle is the foundation for more advanced concepts and practical applications.
How Concentration Gradients Operate: Detailed Steps
To fully grasp how concentration gradients work, it’s essential to understand the process in detail:
Step 1: Identify the Gradient
First, you need to recognize areas of different concentrations. For instance, in a biology context, you might be observing the concentration of a nutrient in various parts of a plant or animal. In a chemical lab, it might involve checking the concentration of reactants in different sections of a solution.
Step 2: Understand Diffusion
Once you've identified a concentration gradient, you need to understand that diffusion occurs. Here's a more in-depth look at the diffusion process:
- Diffusion is the movement of particles from a region of higher concentration to a region of lower concentration.
- This movement continues until equilibrium is reached, where the concentration of particles is equal across the area.
Step 3: Measuring Concentration
To quantify how much diffusion is happening, scientists use concentration measurements. Tools like spectrophotometers can help measure changes in concentration over time in chemical processes, while microscopes combined with sensors can monitor biological concentrations.
Step 4: Apply Knowledge
Now, apply this understanding in practical scenarios:
- In agriculture, knowing how water moves through soil can optimize irrigation methods.
- In medicine, understanding how drugs move through the bloodstream can improve dosing strategies.
By comprehending these steps, you'll be well-equipped to apply the principles of concentration gradients in your own field, whether it’s science, healthcare, or environmental management.
Advanced Understanding: Real-World Examples
To put these concepts into a real-world context, consider these examples:
Example 1: The Role of Gradients in Environmental Science
In environmental science, understanding concentration gradients is crucial for monitoring pollutants. For instance, tracking the concentration gradient of a pesticide in soil and groundwater helps in assessing its impact and determining appropriate cleanup strategies.
Example 2: Pharmaceutical Delivery Systems
In pharmaceuticals, the gradient concept is essential for designing effective drug delivery systems. By understanding the concentration gradient of a drug in the body, scientists can design more efficient pills or injections that ensure the drug reaches the target concentration at the desired rate.
Example 3: Ecosystems and Biodiversity
In ecology, concentration gradients help explain nutrient and pollutant dispersal in ecosystems. Understanding these gradients aids in managing natural resources and protecting biodiversity.
Practical FAQ
What is an example of a concentration gradient in everyday life?
A common example is the diffusion of perfume in a room. When you spray perfume, the concentration is initially high near the source. As it disperses, the concentration decreases throughout the room, following a concentration gradient until the entire room smells of perfume.
How can I measure a concentration gradient in a lab?
To measure a concentration gradient in a lab setting, you would typically use techniques such as chromatography for chemical substances or spectrophotometry. For biological samples, you could use a microscope with a concentration sensor to track changes in concentration over time.
What mistakes should I avoid when studying concentration gradients?
One common mistake is confusing the concept of concentration gradient with the rate of diffusion. The gradient influences but does not determine the speed of diffusion. Also, avoid assuming uniform diffusion rates across all environments without specific evidence.
By integrating these practical examples and detailed steps, you should now have a solid foundation to not only understand but also apply the concept of concentration gradients in real-world scenarios. This knowledge will be instrumental in making informed decisions and innovations in your respective fields.