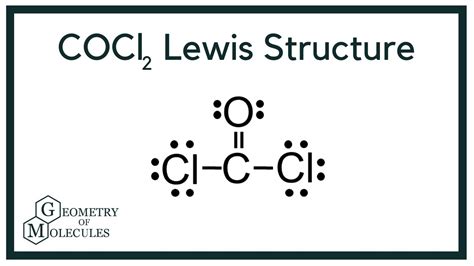
Are you struggling to understand the Lewis structure of COCl₂ and looking for a practical, step-by-step guide? Whether you’re a student facing an exam or a professional delving into advanced chemistry, this guide will walk you through every essential detail. COCl₂, or phosgene, has a unique Lewis structure that, once mastered, will greatly enhance your understanding of molecular geometry and bonding. Here, we break it down to make this complex topic accessible and easy to grasp.
Understanding the COCl2 Lewis Structure: A Comprehensive Guide
To begin with, comprehending the COCl₂ Lewis structure is pivotal for mastering the concept of molecular geometry and bonding. The structure of COCl₂ can be particularly challenging due to its unique bonding patterns and the electron-withdrawing nature of the chlorine atoms. This guide provides an exhaustive exploration to demystify the Lewis structure, ensuring you can apply these insights in real-world scenarios.
Problem-Solution Opening Addressing User Needs
When you’re confronted with complex molecules like COCl₂, deciphering its Lewis structure can feel overwhelming. This comprehensive guide aims to simplify the intricate steps, focusing on your specific needs—whether you’re aiming to ace a chemistry exam, understand industrial applications of phosgene, or simply expand your scientific knowledge. We’ll address your pain points head-on, providing clear, actionable advice, real-world examples, and practical solutions. By the end of this guide, you’ll not only understand the Lewis structure of COCl₂ but also how to approach similar complex molecules with confidence.
Quick Reference
- Immediate action item: Start by drawing the skeleton structure.
- Essential tip: Remember, the least electronegative atom (usually carbon) should be the central atom.
- Common mistake to avoid: Misplacing lone pairs, which is crucial for a stable Lewis structure.
Step-by-Step Guidance for Drawing COCl2 Lewis Structure
Drawing the Lewis structure for COCl₂ involves several key steps. Let’s delve into a detailed explanation to ensure you can master it:
Step 1: Determine the Total Number of Valence Electrons
To begin, calculate the total number of valence electrons available. Carbon © has four valence electrons, each oxygen (O) has six valence electrons, and each chlorine (Cl) atom has seven. Therefore, the calculation is as follows:
Total valence electrons = 4 (from C) + 2 * 6 (from O) + 2 * 7 (from Cl) = 26 valence electrons
Step 2: Draw the Skeleton Structure
Start by drawing the skeleton structure where the carbon atom is at the center, bonded to two oxygen atoms by double bonds, and also bonded to two chlorine atoms by single bonds.
Note: Carbon, being the central atom, will form four bonds. Since there are two oxygen atoms and two chlorine atoms, and each oxygen forms a double bond, this accounts for all four bonds from carbon.
Step 3: Distribute the Remaining Electrons
Now, we distribute the remaining valence electrons to satisfy the octet rule for all atoms. Follow these steps:
- Start by placing lone pairs on the terminal atoms (oxygen and chlorine). Each should get three lone pairs, as they need a total of eight electrons (the octet rule).
- Next, place any remaining electrons on the central atom (carbon). However, note that carbon will only have a sextet (six electrons) due to the double bonds with oxygen.
Step 4: Check for Octet Rule Compliance
Verify that all atoms comply with the octet rule, except for the carbon atom, which has a sextet. This is acceptable due to its central position and the double bonds it forms.
Here’s a quick recap:
- Carbon: Four single bonds (no lone pairs)
- Oxygen atoms: Each with two lone pairs
- Chlorine atoms: Each with three lone pairs
Advanced Considerations: Resonance and Formal Charges
For an in-depth understanding, consider the resonance structures and formal charges:
- Resonance: Although COCl₂ has only one predominant Lewis structure, examining different resonance forms helps in understanding molecule stability.
- Formal Charges: Calculate the formal charge for each atom to ensure it matches the actual charge distribution.
Let’s calculate the formal charge for carbon:
Formal charge on carbon = (valence electrons of C – (bonding electrons – non-bonding electrons)) = 4 – (4 – 0) = 0
Similarly, calculate for oxygen and chlorine atoms:
Formal charge on each oxygen = (6 – (2 – 6)) = 0
Formal charge on each chlorine = (7 – (2 – 6)) = 0
Practical FAQ
Why is the Lewis structure of COCl2 important?
Understanding the Lewis structure of COCl₂ is crucial for grasping its chemical behavior, especially in reactions and industrial applications. It helps predict the molecule’s geometry, reactivity, and how it interacts with other molecules.
How does COCl2 differ in structure from other similar molecules?
COCl₂ differs from similar molecules like CO₂ or CCl₄ due to its specific bonding patterns and the presence of chlorine atoms. CO₂ has a linear structure, while CCl₄ has a tetrahedral geometry. COCl₂’s Lewis structure reveals a bent structure around the carbon due to the double bonds with oxygen and single bonds with chlorine.
Can COCl2 exist in different resonance forms?
While COCl₂ has one predominant structure, exploring possible resonance forms helps understand its stability. However, in practical scenarios, the primary Lewis structure suffices for most chemical discussions.
By following this guide, you’ll gain a thorough understanding of the COCl₂ Lewis structure, addressing both fundamental and advanced aspects. The practical examples, tips, and structured approach ensure you can apply these insights in real-world chemical analyses and beyond.