Unveiling the Molecular Mystery: CO32 Lewis Structure
Understanding the CO32 Lewis structure is essential for grasping the molecular geometry and bonding in carbonates. This guide walks you through the steps required to construct an accurate Lewis structure for the carbonate ion. We'll tackle common challenges you may encounter and offer practical solutions to avoid common pitfalls. Let’s dive into the world of molecular mysteries and decode the CO32 Lewis structure!
Step-by-Step Guidance to Constructing the CO32 Lewis Structure
The carbonate ion (CO32-) consists of one central carbon atom bonded to three oxygen atoms. This structure can appear complex, but breaking it down into manageable steps will make it straightforward. Here’s a detailed, problem-solving approach.
Problem-Solution Opening
Many students find it daunting to draw the Lewis structure of CO32- due to its unique bonding characteristics. Understanding the distribution of electrons, the number of lone pairs, and the formal charges is crucial for accurately representing the molecule. This guide will simplify the process, making it easy for anyone to master the CO32 Lewis structure.
Quick Reference Guide
Quick Reference
- Immediate Action Item: Determine the total number of valence electrons (24 for CO32-).
- Essential Tip: Place the least electronegative atom (Carbon) in the center and arrange the Oxygen atoms around it.
- Common Mistake to Avoid: Forgetting to balance the charges and distribute lone pairs to satisfy the octet rule.
Detailed How-to Sections
Step 1: Counting Valence Electrons
The first step in constructing the CO32 Lewis structure is to determine the total number of valence electrons. Each oxygen atom has 6 valence electrons, and carbon has 4. The ion carries a 2- charge, which means we must add 2 additional electrons to account for the charge:
Carbon: 4 valence electrons
Oxygen: 3 atoms x 6 electrons = 18 electrons
Charge: +2 electrons
Total valence electrons: 4 + 18 + 2 = 24 electrons
Step 2: Setting Up the Skeleton Structure
The next step is to set up the skeleton structure. Place the carbon atom in the center, surrounded by the three oxygen atoms. Carbon forms single bonds with each oxygen atom, using up 3 of the 24 electrons:
- Carbon connects to each oxygen with a single bond.
- Each single bond uses 2 electrons (3 bonds use 6 electrons).
- This leaves us with 18 electrons to distribute.
Step 3: Distributing Remaining Electrons
Distribute the remaining electrons to satisfy the octet rule, starting with the oxygen atoms. Each oxygen atom needs 8 electrons to complete its octet. Since we have 18 electrons left:
Start by adding lone pairs to each oxygen atom:
- Each oxygen atom gets a lone pair of electrons, accounting for 6 electrons (2 electrons per oxygen atom).
- Now we have 12 electrons left to distribute.
Step 4: Checking for Octet Completion
After giving lone pairs to the oxygen atoms, check if all atoms have their octets filled:
- Carbon has 4 electrons (2 double bonds with oxygen atoms).
- Each oxygen atom has 8 electrons (2 from the bond and 6 from lone pairs).
However, since the central carbon atom cannot exceed 8 electrons, we adjust by placing extra electrons on the oxygen atoms:
Convert one lone pair on each oxygen atom into a double bond with the central carbon atom:
- One lone pair becomes a double bond.
- One remaining lone pair per oxygen atom.
Step 5: Ensuring Charge Neutrality
Finally, we ensure the overall charge of the molecule is -2:
- Carbon has 4 electrons.
- Each oxygen atom has an extra electron from converting lone pairs into double bonds:
- 2 electrons per oxygen in double bonds + 6 electrons from lone pairs = 8 electrons per oxygen.
- Sum total = 4 (Carbon) + 3 x 6 (Oxygen lone pairs) + 6 (Double bonds converted) = 24 electrons.
- The extra two electrons account for the -2 charge.
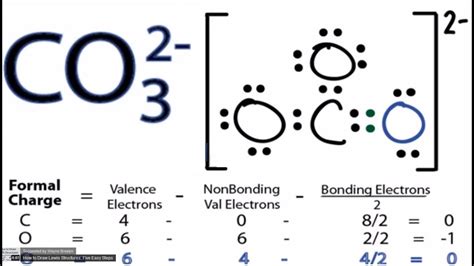
Final CO32- Lewis Structure
The final Lewis structure of CO32- looks like this:
The central carbon atom is double-bonded to two oxygen atoms and single-bonded to one oxygen atom. The double-bonded oxygen atoms have two lone pairs each, while the single-bonded oxygen has three lone pairs.
Practical FAQ Section
What if the initial setup does not balance the formal charges?
When initial setup doesn't balance the charges, revisit the lone pair distribution and double bond conversion:
- Relocate lone pairs from terminal atoms (oxygens) to satisfy octet rules.
- Convert lone pairs to double bonds if necessary, ensuring to follow the least electronegative atom rule.
- Keep checking formal charges after each modification to reach a balanced Lewis structure.
Remember that a correct Lewis structure will have zero net formal charge and distribute all valence electrons correctly.
Why is it important to account for formal charges?
Formal charge calculation helps in determining the most stable Lewis structure. The correct structure will have the smallest possible formal charges, ideally zero. It ensures that the representation accurately reflects the molecule's bonding and electron distribution:
- Reducing formal charges minimizes instability.
- Balanced formal charges indicate a stable molecular configuration.
- Incorrect formal charges may suggest errors in structure or electron placement.
Always recheck formal charges after any modifications to ensure the most stable configuration.
Best Practices and Tips
Here are some best practices and tips to keep in mind while drawing the CO32 Lewis structure:
- Prioritize the Octet Rule: Always strive to satisfy the octet rule for each atom, while being flexible with double and triple bonds when necessary.
- Focus on Resonance Structures: CO32- has resonance, so consider drawing alternative Lewis structures where double bonds shift positions.
- Balance Formal Charges: Ensure that the sum of formal charges equals the overall charge of the ion.
- Use Electron Dots Wisely: Place lone pairs correctly to avoid exceeding octets and ensure stability.
By following these practical steps and tips, you can successfully draw the CO32 Lewis structure and understand the molecule’s bonding characteristics.