Understanding CO2 molecular geometry is essential for both academic and professional chemistry fields. Carbon dioxide, CO2, is a simple yet profoundly influential molecule, fundamental in various applications such as climate science, chemical engineering, and atmospheric studies. This article delves into the intricacies of CO2 molecular geometry with expert insights, practical applications, and real-world examples.
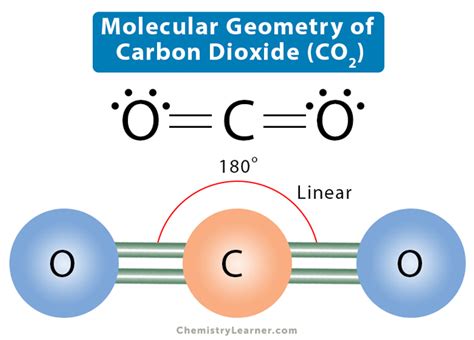
The molecular geometry of CO2 plays a crucial role in its stability and reactivity. This molecule's linear shape, resulting from carbon's double bonds with each oxygen atom, exhibits a symmetry that contributes to its unique properties.
Key Insights
- Primary insight with practical relevance: The linear shape of CO2 is fundamental in determining its role in greenhouse gas dynamics and its chemical inertness.
- Technical consideration with clear application: The symmetric distribution of electrons around the carbon atom leads to a nonpolar molecule, affecting its interactions with other substances.
- Actionable recommendation: For professionals in environmental science, understanding CO2 molecular geometry is pivotal in developing accurate climate models and mitigation strategies.
The Science Behind CO2 Geometry
CO2 molecular geometry is based on the concept of valence shell electron pair repulsion (VSEPR) theory. Carbon dioxide consists of one carbon atom and two oxygen atoms. Carbon, with its four valence electrons, forms two double bonds with each oxygen atom. This arrangement minimizes electron pair repulsion and results in a linear molecular structure, with a bond angle of 180 degrees. The linear geometry implies that the dipole moments of the C=O bonds are equal and opposite, resulting in a molecule that has no net dipole moment.
Significance of CO2 Geometry in Chemistry
The geometry of CO2 has significant implications in various chemical and environmental contexts. The molecule’s linear, nonpolar nature explains its relative inertness under standard conditions, meaning it does not easily react with other substances. This inertness plays a pivotal role in atmospheric chemistry, as CO2 acts as a greenhouse gas that does not readily participate in chemical reactions, unlike other gases like methane or ozone.
Additionally, in industrial processes, the understanding of CO2’s structure helps in designing efficient carbon capture and storage (CCS) technologies. These technologies are crucial for mitigating the impact of CO2 on global warming. Moreover, in biochemical applications, CO2’s geometry and reactivity play roles in processes like photosynthesis, where CO2 is converted into carbohydrates in plants, highlighting the molecule's central role in the carbon cycle.
Question: How does CO2 molecular geometry affect its role in greenhouse gases?
CO2’s linear, nonpolar geometry contributes to its effectiveness as a greenhouse gas by allowing it to remain in the atmosphere for long periods without being readily absorbed or reacted with by other atmospheric components. This prolonged presence leads to increased trapping of heat, thus enhancing the greenhouse effect.
Question: Why is CO2 considered inert in standard conditions?
CO2's molecular geometry, being linear and nonpolar, means that its bonds do not easily dissociate under normal conditions. The stability of the molecule's double bonds with oxygen atoms and the symmetrical electron distribution make CO2 chemically inert, resistant to reactions with other substances.
This detailed exploration of CO2 molecular geometry underscores its importance across diverse fields, from environmental science to industrial applications. By mastering these concepts, professionals can better address the challenges posed by carbon dioxide in our increasingly carbon-dependent world.