Understanding chlorine’s electron configuration is crucial for various scientific fields, from chemistry to environmental science. This knowledge provides a foundation for comprehending chemical bonding, reactivity, and the behavior of chlorine in various chemical processes. The electron configuration of chlorine, specifically, reveals the underlying principles governing its interaction with other elements, thereby influencing everything from industrial applications to ecological concerns.
Key Insights
- Chlorine's electron configuration elucidates its reactivity, particularly its preference for forming chloride ions.
- Understanding electron configuration is crucial for predicting chlorine's behavior in chemical reactions and environmental contexts.
- Identifying chlorine's electron configuration aids in designing effective treatments for water purification and managing pollutants.
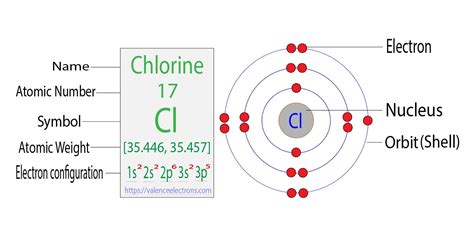
Chlorine’s electron configuration is a direct consequence of its atomic structure. With an atomic number of 17, chlorine has 17 electrons, which are distributed across various orbitals. Its electron configuration is 1s² 2s² 2p⁶ 3s² 3p⁵. This specific arrangement places chlorine in Group 17 (the halogens) of the periodic table, characterized by elements that have seven electrons in their outermost shell.
Understanding this configuration highlights why chlorine atoms readily gain an electron to form chloride ions (Cl⁻). This electron acquisition completes the valence shell, achieving a stable noble gas configuration. The quest for this stability drives chlorine’s reactivity, enabling it to form compounds with metals and non-metals alike. This foundational understanding is pivotal for applications ranging from bleach production to pharmaceuticals and beyond.
Further, chlorine’s electron configuration aids in elucidating its role in various chemical reactions. In oxidation-reduction (redox) reactions, chlorine’s ability to accept an electron and assume a -1 charge is fundamental. This trait is harnessed in numerous industrial processes where chlorine acts as an oxidizing agent. For instance, in water treatment facilities, chlorine is used to disinfect water by targeting harmful microorganisms, thus safeguarding public health.
The reactivity of chlorine also extends to its environmental impact. Chlorine’s electron configuration determines its interaction with organic compounds, often resulting in the formation of toxic byproducts such as dioxins, which pose significant health risks. This insight is critical for developing strategies to mitigate these adverse effects and designing safer chemical processes.
What is the significance of chlorine's electron configuration in environmental science?
Chlorine's electron configuration, specifically its tendency to gain one electron to form Cl⁻, underscores its role in environmental science, particularly in water treatment and pollution control. Understanding this helps in devising effective chlorine use and monitoring its byproducts, which are often harmful pollutants.
How does chlorine's electron configuration influence its reactivity?
Chlorine’s electron configuration reveals its strong tendency to gain an electron to reach a stable octet. This reactivity is key to chlorine's utility as a disinfectant and its involvement in various industrial reactions where it serves as an oxidizing agent.
In summary, the electron configuration of chlorine is fundamental to both its chemical behavior and practical applications. From ensuring the cleanliness of water supplies to playing a role in synthetic organic chemistry, understanding this configuration provides valuable insights into chlorine’s capabilities and limitations, which are essential for advancing both scientific knowledge and technological applications.