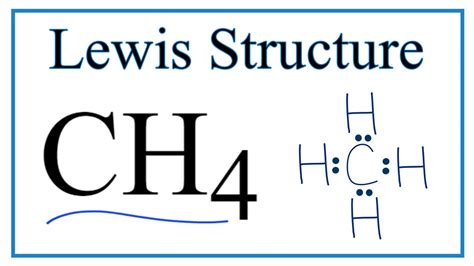
Understanding the intricacies of a methane (CH4) molecule’s Lewis structure is fundamental for any chemistry enthusiast aiming to grasp the basics of molecular bonding. This detailed exploration aims to simplify the complex nature of methane’s structure while ensuring an evidence-based approach.
This guide begins with a foundational overview of the CH4 Lewis structure, a cornerstone concept in chemistry that helps visualize how atoms bond and form molecules. Methane, with its ubiquitous presence in natural gas and as a simple organic compound, provides an ideal platform to delve into molecular geometry and electronic configuration.
Key Insights
- CH4 Lewis structure reveals how carbon forms four single covalent bonds with hydrogen atoms.
- Understanding tetrahedral geometry aids in predicting the molecule's shape and properties.
- This knowledge is foundational for grasping more complex organic molecules.
Understanding Carbon’s Valence Electrons
To unravel the CH4 Lewis structure, we must first examine carbon’s electron configuration. Carbon, located in Group 14 of the periodic table, possesses four valence electrons. These electrons determine its ability to form covalent bonds with other atoms. In the case of methane, carbon seeks to complete its octet by sharing electrons with four hydrogen atoms. This electron-sharing process forms four single covalent bonds, resulting in a stable molecular structure.
Constructing the Lewis Structure
Constructing the CH4 Lewis structure involves several steps grounded in a systematic approach. Initially, we place carbon in the center and surround it with four hydrogen atoms. Carbon shares one electron from each hydrogen atom, forming four single bonds. The Lewis structure can be depicted as:
C in the center, with four H atoms around it, each bonded to C with a single line.
Visualizing this structure reveals its tetrahedral geometry, where each hydrogen atom is positioned equidistant from the others, minimizing repulsion between electron pairs.The tetrahedral arrangement stems from the spatial distribution of the four bonds around the central carbon atom. This is not merely an academic exercise but a real-world application essential for predicting the molecule's behavior, such as its reactivity and physical properties.
Why is understanding the CH4 Lewis structure important?
Understanding the CH4 Lewis structure is vital because it forms the foundation for learning about more complex organic molecules. This knowledge is critical in fields such as chemistry, biochemistry, and material science.
How does the CH4 Lewis structure relate to molecular shape?
The CH4 Lewis structure reveals a tetrahedral molecular shape due to the four single covalent bonds formed between carbon and hydrogen atoms. This geometry minimizes electron pair repulsion, a fundamental concept in molecular chemistry.
In conclusion, the CH4 Lewis structure exemplifies the principles of covalent bonding and molecular geometry. By mastering this concept, chemistry enthusiasts can unlock a deeper understanding of the molecular world, paving the way for advanced studies in organic chemistry and related fields. This rigorous yet approachable exploration serves as an essential stepping stone for anyone eager to delve into the fascinating realm of molecular science.