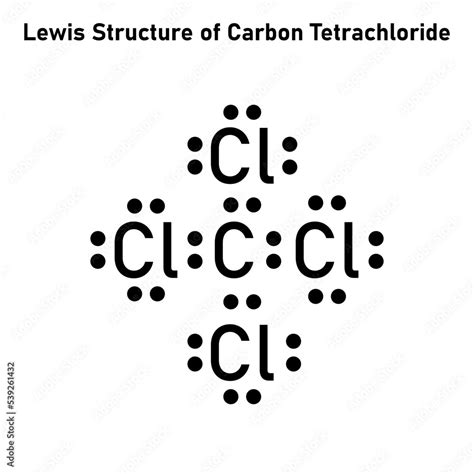
Mastering CCL4 Lewis Structure: Unlock the Secrets of Carbon Tetrachloride Bonding
The carbon tetrachloride molecule (CCL4) has been widely studied due to its significant industrial applications and environmental relevance. Understanding its Lewis structure is crucial for comprehending its chemical behavior, bonding nature, and the implications of its molecular geometry. Here, we will delve into the intricacies of the CCL4 Lewis structure, offering insights backed by evidence and real-world examples to ensure a comprehensive understanding.
Key Insights
- Carbon tetrachloride (CCL4) is a tetrahedral molecule with four equivalent C–Cl bonds.
- The central carbon atom in CCL4 forms four single bonds with four chlorine atoms.
- It is essential to apply the principles of VSEPR theory for understanding its tetrahedral geometry.
Understanding the Basics of Lewis Structures
In chemistry, the Lewis structure helps visualize the arrangement of atoms and electrons in a molecule. A Lewis structure provides a simple representation of valence electrons of an atom that participate in the bond formation. For CCL4, the Lewis structure reveals how the central carbon atom shares its electrons with four chlorine atoms. Carbon, with its four valence electrons, can form four covalent bonds to achieve a stable electron configuration, similar to that of a noble gas. This is complemented by each chlorine atom, which has seven valence electrons and seeks one more electron to achieve stability. The resultant structure is a perfect tetrahedral arrangement with carbon at the center.Decoding the Tetrahedral Geometry of CCL4
To grasp the molecular geometry of CCL4, one must apply the Valence Shell Electron Pair Repulsion (VSEPR) theory. According to VSEPR, electron pairs around a central atom repel each other and arrange themselves to minimize repulsion. In CCL4, there are four bonded pairs of electrons around the central carbon atom, leading to a tetrahedral geometry. Each chlorine atom is positioned at the vertices of a tetrahedron, with the carbon atom at the center. This arrangement effectively minimizes electron pair repulsion, resulting in a stable molecular configuration. The bond angle in a perfect tetrahedral molecule is 109.5 degrees, which holds true for CCL4.What is the significance of CCL4 in industrial applications?
Carbon tetrachloride (CCL4) has been extensively used as a solvent in various industrial processes, including as a precursor in the production of chlorofluorocarbons (CFCs). It also served as a cleaning agent and in the manufacturing of refrigerants. However, due to its harmful health and environmental effects, its use has significantly declined.
How does CCL4 affect the environment?
CCL4 has been shown to persist in the environment and bioaccumulate, leading to significant ecological and health impacts. It is known to cause liver and kidney damage in humans and is also a suspected carcinogen. Consequently, it has been phased out in many applications to mitigate its harmful effects.
This deep dive into the Lewis structure and geometry of CCL4 provides a critical understanding of its bonding characteristics and molecular behavior. By mastering this fundamental aspect of CCL4, one can appreciate its broader chemical significance and the imperative of understanding such structures in the context of modern chemical applications and environmental stewardship.