Mastering Carbon Disulfide (CS₂) Lewis Structure Secrets
Understanding the Lewis structure of carbon disulfide (CS₂) is essential for chemists as it reveals the molecule’s stability, reactivity, and bonding nature. This knowledge can be directly applied to predict chemical behavior, which is pivotal in fields like organic chemistry and material science. Carbon disulfide is a linear molecule, which has significant implications for its physical properties and reactivity.
Key insights
Key Insights
- Carbon disulfide (CS₂) has a linear molecular geometry which is critical for its reactivity and physical properties.
- Understanding the double bond between carbon and sulfur atoms helps in grasping the molecule’s bonding nature.
- Proper depiction of lone pairs is essential to fully grasp the molecule’s electron distribution.
Analysis of Carbon Disulfide’s Bonding Nature
Bonding Characteristics of CS₂
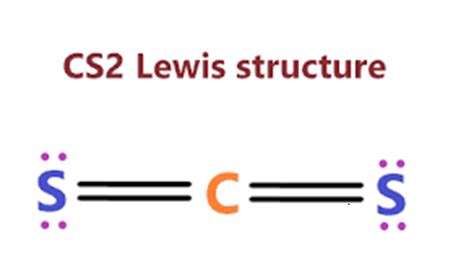
The structure of carbon disulfide is dominated by its linear shape, which stems from the central carbon atom’s sp hybridization. This hybridization allows the carbon atom to form two double bonds with the two sulfur atoms, creating a molecule with a 180° angle between them. Each double bond consists of a sigma bond and a pi bond. The pi bond formation is crucial for the molecule’s stability and affects its reactivity, particularly towards nucleophiles and electrophiles.
To draw the Lewis structure, begin with the central carbon atom surrounded by the two sulfur atoms. Carbon has four valence electrons, and each sulfur atom contributes six. To satisfy the octet rule, carbon forms two double bonds with each sulfur. This accounts for 16 valence electrons in total. However, to achieve full octet satisfaction for the sulfur atoms, each sulfur shares an electron pair with carbon, leaving each sulfur with a double bond (two pairs of electrons) directly attached to carbon. This results in no lone pairs on carbon but two lone pairs on each sulfur atom, reflecting a total of four lone pairs distributed around the molecule.
Electronegativity and Resonance Structures
Carbon disulfide also exhibits a unique electronegativity aspect. Sulfur, being less electronegative than carbon, pulls electron density from the carbon atom. This differential electron density, though minimal, can have observable effects in chemical reactions. Additionally, despite being a simple molecule, CS₂ does not possess resonance structures, which simplifies its analysis but also limits the extent of electron density distribution across the molecule. Unlike compounds that show resonance, CS₂’s Lewis structure is straightforward, which helps in simplifying its reactivity and chemical interactions.
FAQ section
What is the geometry of the CS₂ molecule?
The geometry of the CS₂ molecule is linear. This linear geometry arises from the sp hybridization of the central carbon atom, which forms two double bonds with the sulfur atoms.
Is there any resonance in carbon disulfide?
No, there is no resonance in carbon disulfide (CS₂). This simplification means that the Lewis structure accurately represents the electron distribution within the molecule.
Understanding the Lewis structure of carbon disulfide is not merely an academic exercise but a critical tool in predicting the molecule’s behavior in various chemical contexts. The simplicity and straightforward nature of CS₂ make it an excellent study subject for grasping foundational concepts in molecular chemistry.