Calcium carbonate, often abbreviated as CaCO3, is an abundant compound, especially noted in various industries ranging from pharmaceuticals to construction. Understanding its molar mass is crucial for applications like chemical synthesis, nutritional labeling, and environmental studies. This article aims to demystify the molar mass of CaCO3 in a straightforward manner, providing expert insights and practical examples for a clear understanding.
Key Insights
- Primary insight with practical relevance: Calculating the molar mass of CaCO3 is essential for stoichiometric calculations in chemical reactions.
- Technical consideration with clear application: Accurate knowledge of CaCO3 molar mass enables precise formulation of products in industries like food and construction.
- Actionable recommendation: Use the periodic table to determine the molar mass by summing atomic masses of calcium (Ca), carbon (C), and three oxygen (O) atoms.
Decomposing the Atomic Composition
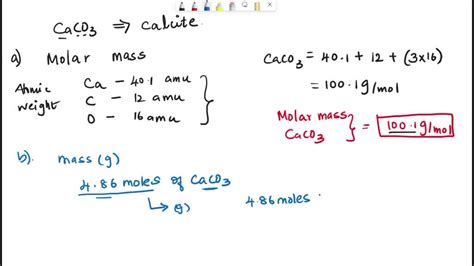
To find the molar mass of CaCO3, one must consider the atomic masses of its constituent elements: calcium, carbon, and oxygen. Calcium has an atomic mass of approximately 40.08 g/mol, carbon is around 12.01 g/mol, and oxygen weighs about 16.00 g/mol per atom. Since there are three oxygen atoms in the compound, the calculation should account for 3 times the atomic mass of oxygen. Adding these values together provides the total molar mass:
Molar mass of CaCO3 = 40.08 g/mol (Ca) + 12.01 g/mol © + 3 * 16.00 g/mol (O) = 100.09 g/mol
Applications in Various Industries
Understanding the molar mass of CaCO3 is invaluable in many fields. For instance, in the pharmaceutical industry, precise molar mass calculations ensure accurate dosages in calcium supplements. In agriculture, calcium carbonate is used as a soil conditioner, and its molar mass helps in determining the correct quantities for pH adjustment. In construction, CaCO3 is a fundamental component in cement and mortar, where its molar mass contributes to the understanding of mixing ratios and formulations. For environmental studies, knowing the molar mass helps in assessing the impact of calcium carbonate in biomineralization and carbon sequestration.
What is the significance of knowing the molar mass of CaCO3?
Knowing the molar mass is critical for precise chemical calculations and formulations across various industries including pharmaceuticals, agriculture, construction, and environmental studies.
How does CaCO3 molar mass apply in dietary supplements?
In dietary supplements, precise molar mass calculations ensure the accurate delivery of calcium, which is essential for bone health and other physiological functions.
By grasping the concept of molar mass and its applications in practical scenarios, professionals and students alike can leverage this knowledge to enhance their work efficiency and precision. The clear understanding of CaCO3 molar mass enables more effective and safer use in scientific and industrial processes.