Are you struggling to understand the hidden structures within bent geometry molecules? If so, you’re not alone. Bent geometry can be tricky, but fear not! This guide is designed to help demystify these molecular shapes and provide you with clear, practical solutions to grasp their structure and behavior.
Understanding Bent Geometry Molecules
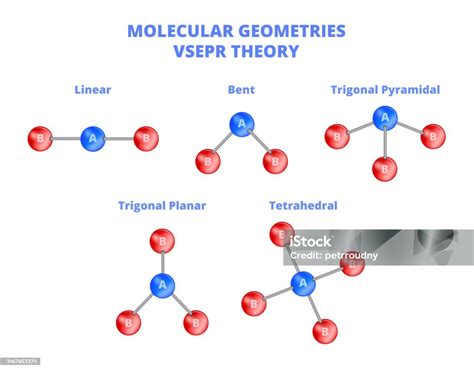
Bent geometry, also known as V-shaped or angular geometry, occurs in molecules where a central atom is bonded to two or more atoms but has a lone pair of electrons. These lone pairs repel the bonded pairs, resulting in a bent structure. A classic example is water (H2O). Understanding this shape is crucial because it impacts molecular polarity, reactivity, and physical properties.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Start identifying the central atom and lone pairs to visualize the bent structure.
- Essential tip with step-by-step guidance: Use the VSEPR (Valence Shell Electron Pair Repulsion) theory to predict molecular shapes accurately.
- Common mistake to avoid with solution: Overlooking lone pairs which can significantly distort the molecular geometry.
How to Understand and Apply Bent Geometry
The journey to understanding bent geometry starts with a fundamental grasp of molecular bonding and electron pair repulsion. Here’s a detailed guide to help you:
Step-by-Step Guide to Bent Geometry
Here’s how to determine and understand bent geometry:
- Identify the Central Atom: Determine which atom in the molecule is central. Typically, it is the least electronegative atom that forms bonds with other atoms.
- Count the Electron Pairs: Count the bonding pairs and lone pairs around the central atom. For example, in water (H2O), oxygen has two bonded pairs to hydrogen atoms and two lone pairs.
- Use VSEPR Theory: Apply the Valence Shell Electron Pair Repulsion theory. Lone pairs repel more strongly than bonding pairs, pushing bonded atoms into a bent arrangement. The bent shape minimizes repulsion and stabilizes the molecule.
To make this more concrete, let’s examine water:
- Central Atom: Oxygen (O) is the central atom.
- Electron Pairs: Oxygen has two lone pairs and two bonding pairs with hydrogen atoms.
- Repulsion: The two lone pairs and two bonding pairs arrange themselves to minimize repulsion, forming a bent shape with an H-O-H angle of approximately 104.5 degrees.
Advanced Techniques for Analyzing Bent Molecules
Once you’ve mastered the basics, advanced techniques can further refine your understanding of bent geometry molecules:
- Hybridization: Understand the concept of hybrid orbitals. In bent molecules like water, the central atom (oxygen) often undergoes sp3 hybridization, allowing the formation of four equivalent orbitals (two for lone pairs and two for bonding pairs).
- Molecular Orbitals: For more complex molecules, visualize molecular orbitals to understand electron distribution and bond formation.
- Physical Properties: Learn how bent geometry affects physical properties like boiling and melting points, and solubility. For instance, molecules with bent geometry often have stronger intermolecular forces if they are polar.
Tips and Best Practices
Here are some practical tips to keep in mind when studying bent geometry molecules:
- Practice Visualization: Use 3D molecular models or software to visualize the shapes. Physical models can be extremely helpful.
- Review Common Examples: Familiarize yourself with common bent molecules like water, sulfur dioxide (SO2), and phosphorus trichloride (PCl3).
- Check Your Work: Always double-check your work, especially when counting electron pairs and applying VSEPR theory.
Practical FAQ
What is the difference between bent and V-shaped molecules?
The terms ‘bent’ and ‘V-shaped’ are often used interchangeably. Both refer to molecules where the central atom has a lone pair and two bonding pairs, resulting in a bent shape. This structure is commonly observed in molecules like water (H2O). While ‘V-shaped’ can imply a more simplified view, ‘bent’ often emphasizes the angular nature influenced by the lone pairs.
How does bent geometry affect molecular polarity?
Bent geometry typically results in a polar molecule due to the uneven distribution of electron density caused by the lone pairs and the angles between bonded pairs. In water, for example, the uneven charge distribution creates a dipole moment, leading to polar properties such as high boiling points and strong hydrogen bonding capabilities.
Can bent geometry lead to isomerism?
Yes, bent geometry can lead to structural isomerism, where different compounds have the same molecular formula but different arrangements of atoms. However, since bent geometry usually arises from specific electronic configurations, it’s less common for isomers with the same central atom and electron count to have bent geometry. For instance, cis and trans isomers in other geometries could exist depending on the molecular structure.
Through these steps, tips, and examples, you’ll gain a comprehensive understanding of bent geometry molecules, allowing you to tackle related problems with confidence. Whether you’re tackling exams, conducting research, or exploring chemistry as a hobby, this guide provides the foundation you need to dive deep into molecular shapes and their implications.