Decoding the BCl3 Lewis Structure: Unveil Key Chemistry Insights
Understanding the BCl3 (boron trichloride) Lewis structure can provide significant insight into the behavior and properties of this compound. Whether you’re studying chemistry to grasp fundamental concepts or aiming to dive into more advanced topics, mastering this structure will lay a solid foundation. Here, we will guide you through the process of decoding the BCl3 Lewis structure with actionable advice and practical examples to ensure clarity and comprehension.
Problem-Solution Opening Addressing User Needs
Many students find the concept of drawing Lewis structures daunting, particularly for compounds like BCl3 where the geometry and electron distribution can be confusing. Understanding the Lewis structure is crucial for grasping molecular geometry, bonding, and reactivity. This guide is designed to break down the BCl3 Lewis structure into understandable and manageable steps, providing you with real-world applications of your newfound knowledge. Whether you're a high school student, a college beginner, or an advanced learner looking to refresh, this guide will equip you with the necessary tools to decode BCl3’s structure efficiently.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Always start by calculating the total valence electrons. For BCl3, boron has 3 valence electrons and each chlorine atom has 7, making a total of 24 valence electrons.
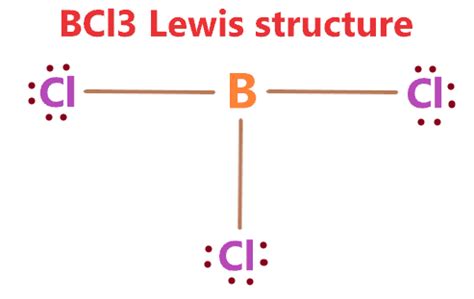
- Essential tip with step-by-step guidance: Place the least electronegative atom in the center—in BCl3, boron is the central atom surrounded by three chlorine atoms.
- Common mistake to avoid with solution: Do not neglect to draw lone pairs; boron needs only three electrons from chlorine atoms and none on itself, so there are no lone pairs on boron.
Detailed How-To Sections
Understanding Valence Electrons
To begin with the BCl3 Lewis structure, it’s important to grasp the concept of valence electrons. Valence electrons are the outermost electrons of an atom and are involved in chemical bonding. Here’s how we determine the valence electrons for BCl3:
- Boron has 3 valence electrons.
- Each chlorine atom has 7 valence electrons.
- Therefore, for BCl3, we calculate the total valence electrons as follows:
Total valence electrons = (3 from Boron) + (3 * 7 from three Chlorine atoms) = 3 + 21 = 24 valence electrons
Constructing the Initial Skeleton
Next, we need to create a skeletal structure for BCl3. Here’s how to do it:
1. Place the boron atom in the center and the three chlorine atoms around it. This is because boron is less electronegative compared to chlorine, thus it should be in the center.
2. Draw single bonds from boron to each of the three chlorine atoms. This uses up 6 valence electrons (3 bonds * 2 electrons per bond) leaving 18 electrons to distribute:
Assigning Remaining Electrons
Now we need to assign the remaining electrons to fulfill the octet rule, which states that atoms tend to gain, lose, or share electrons to have a full set of eight valence electrons.
However, for BCl3, boron does not require any additional electrons from the chlorine atoms because it shares three electrons with each chlorine atom through the single bonds, forming a total of three covalent bonds:
In BCl3, boron has only six electrons around it, yet it is stable because of the trigonal planar geometry that allows boron to achieve stability by utilizing its p orbitals effectively. The remaining electrons (18) are distributed as follows:
12 electrons are assigned to the chlorine atoms as lone pairs (each chlorine gets three lone pairs, totaling 6 lone pairs distributed to three chlorine atoms).
Reviewing the Lewis Structure
Finally, review the structure:
1. Confirm that boron, being in group III, shares three electrons to form three bonds, thus requiring a total of six electrons (3 bonds).
2. Confirm that each chlorine atom has eight electrons (three lone pairs and one from the bond).
3. Double-check that all 24 valence electrons have been used:
- 3 bonds * 2 electrons = 6 electrons
- 6 lone pairs on chlorine * 3 chlorine atoms * 2 electrons per pair = 12 electrons
- Total electrons: 6 + 12 = 18 electrons used
- Boron's remaining electrons = 6
- Total = 24 valence electrons
Practical Insights and Applications
Beyond the theoretical understanding, knowing the BCl3 Lewis structure can help in practical applications:
1. Reactivity and Chemical Behavior: BCl3 is a trigonal planar molecule which affects its chemical reactivity. The geometry means that boron has no lone pairs to disturb its electron cloud, making it more stable and less likely to react spontaneously.
2. Industrial Uses: BCl3 is used in semiconductor manufacturing and as a precursor in the production of boron compounds.
3. Environmental Impact: Knowledge of BCl3’s structure aids in understanding its behavior in environmental contexts, including its potential toxicity and interactions with other molecules.
Practical FAQ
Common user question about practical application
What is the significance of the BCl3 Lewis structure in chemical reactions?
Understanding the BCl3 Lewis structure is essential because it reveals the molecule’s geometry and reactivity. The trigonal planar shape of BCl3, derived from its Lewis structure, means that it has a symmetrical arrangement that impacts how it interacts with other substances. For instance, in reactions where BCl3 might act as a Lewis acid, the understanding of its structure aids in predicting its behavior in accepting electron pairs from other molecules.
Best Practices for Mastering Lewis Structures
To enhance your understanding and retention of Lewis structures, consider the following best practices:
- Practice regularly by drawing different molecules, starting with simple ones like water (H2O) and gradually moving to more complex ones like BCl3.
- Use mnemonic devices to remember valence electron counts for common elements (e.g., “Group 1 has one, Group 2 has two, Group 13 has three...”).
- Use colored pencils or markers to distinguish between different elements when drawing structures, which can make visualizing connections clearer.
- Review and practice the octet rule and exceptions to it, as understanding when and why atoms may deviate from this rule is key to mastering Lewis structures.
- Always double-check your work by ensuring all valence electrons are accounted for and correctly placed.
By following these steps and tips, you can confidently decode the BCl3 Lewis structure and apply this knowledge to other molecules, making you well-equipped in your chemistry studies.