The question of when exactly water freezes often eludes even the most seasoned scientists. It’s a deceptively simple query, yet its answer requires diving into the depths of thermodynamics and molecular behavior. Understanding this transformation is vital not just for scientific curiosity but also for practical applications such as climate science, engineering, and everyday life.
Key Insights
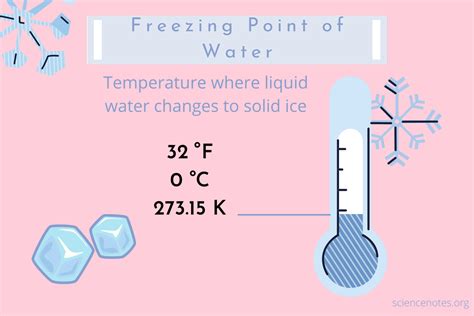
- Water freezes at 0°C (32°F) under standard atmospheric pressure.
- Pressure changes and impurities can affect freezing points.
- Knowing these factors can help in practical applications such as preserving food or designing infrastructure in cold climates.
Thermodynamics of Freezing
When we talk about water freezing, we delve into the world of phase changes. Water is a molecule composed of hydrogen and oxygen, and its behavior under different temperatures is governed by thermodynamic laws. At 0°C (32°F), water transitions from a liquid to a solid state, a process known as freezing. This transition occurs because the thermal energy provided by the surrounding environment decreases until it’s insufficient to sustain the liquid state. Below this temperature, water molecules lose kinetic energy and arrange themselves into a crystalline structure—ice.Environmental Influences
Though 0°C (32°F) marks the standard freezing point for pure water under one atmosphere of pressure, environmental factors can alter this. Pressure changes, for instance, can elevate or reduce the freezing point. This phenomenon is crucial in high-altitude areas where the atmospheric pressure is lower, causing water to freeze at temperatures slightly below 0°C (32°F). Conversely, in deeper ocean waters, increased pressure can raise the freezing point. Furthermore, impurities such as salt, sugars, or other solutes lower the freezing point—a principle widely used in refrigeration to prevent ice crystal formation in frozen foods.In a real-world context, knowing these environmental influences allows for precise control in processes like freezing food. For instance, the addition of glycerol to ice cream mix lowers the freezing point, resulting in a smoother texture and preventing large ice crystals that can ruin the mouthfeel.
Does altitude affect the freezing point of water?
Yes, altitude can affect the freezing point of water. At higher altitudes, the atmospheric pressure is lower, which typically causes water to freeze at temperatures slightly below 0°C (32°F).
How does salt affect the freezing point of water?
Salt lowers the freezing point of water due to a colligative property. When salt is mixed with water, it disrupts the hydrogen bonding structure, causing the water to freeze at a lower temperature. This effect is utilized in de-icing salt spread on icy roads.
This nuanced understanding of the freezing point of water equips us with practical tools to manage various scientific and everyday challenges, highlighting the importance of foundational scientific knowledge in real-world applications. From food preservation to designing infrastructure in polar regions, comprehending the precise conditions under which water freezes is a cornerstone of many technological advancements.