Anti Markovnikov Rule Explained: Surprising Facts in Organic Chemistry
If you’re studying organic chemistry, you’ve likely encountered the Markovnikov’s rule, which is the standard guideline for predicting the outcomes of certain addition reactions. However, it’s equally important to understand when and why the Anti Markovnikov Rule comes into play, as it offers an alternative path to similar reactions, often surprising those new to the field with its nuances.
Understanding both rules can provide a deeper appreciation of the complexities and variations in organic chemical reactions, helping you anticipate and manipulate reaction pathways more effectively. In this guide, we'll dive deep into the Anti Markovnikov Rule, offering step-by-step guidance, practical solutions, and addressing common pain points you may encounter.
Quick Reference
Quick Reference
- Immediate action item: When dealing with an unsymmetrical alkene and HBr, consider the possibility of the Anti Markovnikov addition if a peroxide is present.
- Essential tip: To achieve Anti Markovnikov addition, use a radical initiator like a peroxide which breaks the reaction pathway from the traditional route.
- Common mistake to avoid: Don’t assume Markovnikov’s rule always applies; always check for conditions like peroxides or catalytic amounts of acids.
Understanding the Anti Markovnikov Addition
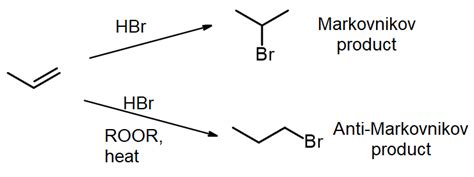
The Anti Markovnikov rule predicts the opposite outcome compared to Markovnikov’s rule in hydrogen halide addition to unsymmetrical alkenes. Typically, Markovnikov’s rule dictates that the hydrogen attaches to the carbon with the most hydrogens, while the halide attaches to the carbon with fewer hydrogens. Conversely, the Anti Markovnikov addition leads to the hydrogen attaching to the carbon with fewer hydrogens, and the halide to the carbon with more hydrogens.
The most commonly seen occurrence of the Anti Markovnikov addition is facilitated by the presence of peroxides. This happens because peroxides initiate radical reactions that diverge from the conventional carbocation mechanism that Markovnikov’s rule follows.
Step-by-Step Guide to Performing Anti Markovnikov Addition
Here’s how to guide your reactions to follow the Anti Markovnikov pathway.
- Identify the Reaction: Determine if the addition reaction of an alkene with a hydrogen halide will likely follow the Anti Markovnikov rule. This occurs primarily with unsymmetrical alkenes in the presence of a radical initiator like a peroxide.
- Prepare the Reaction Mixture: If peroxides are involved, they are usually added in catalytic amounts to ensure radical initiation. Prepare a mixture that includes your unsymmetrical alkene and the hydrogen halide (e.g., HBr or HCl).
- Initiate the Radical Reaction: Heat the mixture mildly to activate the peroxide. This gentle heating is enough to start the radical chain reaction without causing decomposition. The radical attack typically starts on the carbon with fewer hydrogens.
- Monitor the Reaction: Observe the progress of the reaction, which is slower than ionic addition reactions. The product forms through a radical mechanism rather than through a carbocation intermediate.
- Purification of Product: Once the reaction is complete, employ standard workup procedures such as washing with water and using organic extraction techniques to isolate the product.
To see this in action, consider the addition of HBr to 2-methyl-1-butene:
Under normal conditions, the expected product would be 2-bromo-2-methylbutane following Markovnikov’s rule. However, with a peroxide present, the product will be 1-bromo-2-methylbutane, adhering to the Anti Markovnikov rule.
Detailed Practical Example
Let’s walk through a detailed example to understand how this works in practice. Here, we will add HBr to 1-pentene in the presence of a peroxide:
- Step 1: Start with 1-pentene, the unsymmetrical alkene, and a small amount of a peroxide, such as tert-butyl peroxide.
- Step 2: Heat the mixture gently. The peroxide will break down to form radicals:
- Peroxide → 2 RO•
- RO• + HBr → ROH + Br•
- Step 3: The bromine radical (Br•) is highly reactive and will attach to the less substituted carbon of 1-pentene:
- Br• + CH2=CH-CH2-CH2-CH3 → Br-CH2-CH=CH-CH2-CH3
- Step 4: The radical now reacts with another HBr molecule to form a hydrogen bond with the more substituted carbon:
- Br-CH2-CH=CH-CH2-CH3 + HBr → Br-CH2-CH(H)-CH2-CH2-CH3
The final product, 1-bromo-pentene, illustrates the Anti Markovnikov addition.
Practical FAQ
What conditions favor Anti Markovnikov addition?
The Anti Markovnikov addition typically occurs in the presence of a radical initiator, most commonly a peroxide. This is because peroxides help initiate a radical reaction rather than the traditional ionic pathway followed in standard addition reactions. Therefore, any reaction conditions that allow the formation of radicals—such as the presence of a peroxide, heat, or light—favor this addition pattern.
Why doesn’t Anti Markovnikov addition occur without peroxide?
Without peroxide, the addition reaction follows the Markovnikov pathway because the initial mechanism involves carbocation intermediates formed from the protonation of the alkene. In the absence of a radical initiator, the electrophilic addition mechanism dominates, leading to the hydrogen attaching to the more substituted carbon.
How can I confirm the outcome of an Anti Markovnikov addition?
To confirm the outcome, perform an NMR (Nuclear Magnetic Resonance) spectroscopy analysis of the product. This will allow you to determine the position of the substituents and confirm whether the Anti Markovnikov addition has occurred. You can also compare the melting point, boiling point, or other physical properties of the synthesized product with those of known compounds.
This guide provides a comprehensive approach to understanding and performing Anti Markovnikov addition reactions, ensuring that you can effectively manipulate these reactions to achieve the desired products in organic synthesis. With this knowledge, you’ll be equipped to navigate the complexities of organic chemistry reactions with confidence.