Ammonia, NH₃, is a fundamental molecule in both industrial chemistry and biological processes. Understanding the ammonia ion charge is essential for various applications, from fertilizers to understanding protein structure in biochemistry. To truly grasp the concept, we will delve into its chemical behavior, practical implications, and technical nuances.
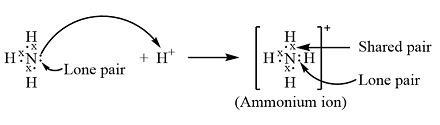
When discussing ammonia and its ion charge, it's vital to understand its behavior in different environments. Ammonia acts as a weak base in aqueous solutions, readily accepting a proton (H⁺) to form the ammonium ion (NH₄⁺). The overall charge of the ammonium ion is +1. This characteristic is pivotal in various fields such as agriculture, where ammonium salts serve as essential nitrogen sources for plants.
Key Insights
- Ammonia acts as a weak base, forming the ammonium ion (NH₄⁺) in solution.
- The ammonium ion has a technical importance in creating fertilizers.
- Understanding the charge helps in optimizing its use in agricultural and industrial applications.
Chemical Behavior of Ammonia
Ammonia’s role as a base is rooted in its molecular structure. It has a lone pair of electrons on the nitrogen atom, which it can readily donate to an acid (proton donor). This property is pivotal when ammonia interacts with water. In a solution, ammonia reacts with water as follows:
NH₃ + H₂O ⇌ NH₄⁺ + OH⁻
This reaction produces both the ammonium ion and hydroxide ions, illustrating ammonia’s basic nature. This process is reversible, meaning the extent of ionization can be influenced by factors such as temperature and concentration.
Industrial and Agricultural Applications
The practical application of ammonia ion charge is evident in the creation of fertilizers. When ammonium salts like ammonium nitrate (NH₄NO₃) are used, the ammonium ion contributes directly to plant nutrition. Its +1 charge allows it to balance negative charges in soil and provides a critical nutrient for growth. Moreover, the technical consideration here is the stability and solubility of ammonium salts, which are optimized to maximize nutrient delivery and minimize environmental impact.
How does the charge of the ammonium ion affect its solubility?
The charge of the ammonium ion influences its solubility in water, leading to the formation of soluble ammonium salts that are crucial for fertilizers. The +1 charge of NH₄⁺ allows it to form stable compounds with anions such as nitrate, sulfate, or phosphate, which enhances their availability in soil.
Why is understanding the ammonia ion charge important for environmental science?
Understanding the charge and behavior of ammonia and its ions is crucial for assessing its environmental impact. Excessive use of ammonia-based fertilizers can lead to leaching, where ammonium ions seep into groundwater, causing eutrophication in water bodies. Recognizing these dynamics helps in developing regulations and best practices to mitigate adverse environmental effects.
In summary, the ammonia ion charge is a central concept in multiple scientific and industrial applications. From basic chemical behavior to significant practical uses in agriculture, a comprehensive understanding of the ammonium ion’s role enhances efficiency and minimizes negative impacts.