Understanding the subtle differences between aldose and ketose sugars can be pivotal for both culinary enthusiasts and those with a scientific curiosity. These two classes of carbohydrates are fundamental in various biological processes and have distinct roles in food chemistry and biochemistry. This guide delves deep into the distinctions, similarities, and practical applications of aldoses and ketoses, providing actionable insights to enrich your knowledge and improve your understanding of these essential sugars.
Problem-Solution Opening Addressing User Needs
If you’ve ever wondered why certain sugars behave differently in recipes or why your understanding of biochemistry isn’t as clear as you hoped, you’re not alone. The difference between aldoses and ketoses often lies beneath the surface of everyday observations and scientific studies. For instance, the sweetness, solubility, and chemical reactivity vary significantly, influencing everything from food preparation to medical treatments. This guide aims to demystify these sugars by offering practical solutions to common questions and misconceptions. By the end of this guide, you’ll have a clearer, actionable understanding of how to use this knowledge in both everyday and advanced contexts.
Quick Reference
Quick Reference
- Immediate action item: Identify the type of sugar in your recipes (aldose or ketose) and understand its impact on flavor and texture.
- Essential tip: Always consider the structural configuration when determining the reactivity and solubility of sugars in your cooking processes.
- Common mistake to avoid: Don’t confuse the chemical behavior of aldoses and ketoses; understanding their differences is key to successful culinary and biochemical applications.
Detailed How-To Section: Aldose Sugars
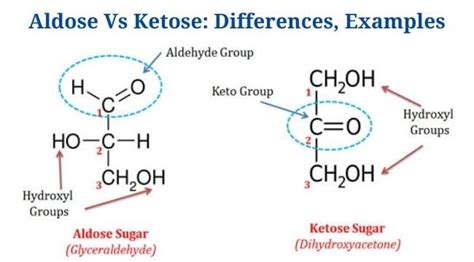
Aldose sugars are a class of monosaccharides containing an aldehyde group at the first carbon atom. They play a significant role in both human nutrition and various metabolic pathways.
Here’s a detailed breakdown of aldose sugars:
To start, let’s dive into the basics of aldoses:
- Identification: Aldose sugars can be identified by the presence of an aldehyde group. Common examples include glucose, galactose, and tagatose.
- Reactivity: Aldehydes are generally more reactive than ketones, leading to a higher likelihood of forming glycosidic bonds in various biochemical processes.
Here’s how you can practically apply this understanding:
- Culinary Use: In cooking, aldose sugars tend to caramelize more easily than ketoses. For instance, when baking, using an aldose sugar like glucose will create a deeper, more intense caramelization.
- Biochemical Insight: In metabolic pathways, aldoses are often involved in glycolysis, where they are broken down to produce energy. Understanding this can help in tailoring dietary choices for optimal health.
To gain more advanced insights:
- Chemical Synthesis: For those involved in advanced biochemistry or pharmaceutical development, aldoses are crucial in the synthesis of more complex sugars and compounds.
- Research Application: In research, knowing how to manipulate aldose sugars can lead to significant advancements in understanding metabolic diseases and developing new therapeutic agents.
Detailed How-To Section: Ketose Sugars
Ketose sugars are another class of monosaccharides, distinguished by a ketone group at the second carbon atom. Unlike aldoses, ketoses include sugars like fructose, ribose, and xylulose, and they play a different role in biochemical processes.
Let’s explore the fundamentals of ketoses:
- Identification: Ketoses can be identified by the presence of a ketone group. Common examples include fructose, found in fruits, and ribose, an essential component of RNA.
- Reactivity: Ketoses tend to have a slower rate of glycosidic bond formation compared to aldoses, influencing their behavior in metabolic and culinary contexts.
Here’s how you can practically apply this understanding:
- Culinary Use: Ketose sugars like fructose are known for their unique sweetness and are often used in recipes to enhance sweetness without caramelization. They’re ideal for low-calorie sweetening options.
- Biochemical Insight: In cellular respiration, ketoses such as fructose are essential in the pentose phosphate pathway, which generates NADPH and ribose-5-phosphate, crucial for nucleotide synthesis.
To gain more advanced insights:
- Chemical Synthesis: Advanced research on ketoses can lead to better understanding of metabolic pathways and diseases, including those related to fructose metabolism.
- Pharmaceutical Development: For drug developers, ketose sugars offer pathways to synthesize new therapeutic agents due to their unique structural properties.
Practical FAQ
Common user question about practical application
How can I tell if a sugar is an aldose or ketose when I’m cooking?
Determining the type of sugar without lab equipment can be tricky, but there are a few clues you can observe:
Flavor and Texture: Aldose sugars like glucose are more likely to caramelize and create a deeper flavor, while ketoses such as fructose dissolve quickly and offer a different kind of sweetness.
Culinary Usage: If a recipe calls for something that caramelizes well, it’s likely an aldose. For recipes where you want a quick dissolve and unique sweetness, like in many fruit-based recipes, the sugar is probably a ketose.
Understanding these differences can greatly enhance your cooking techniques and the final outcome of your recipes.
By grasping these fundamentals and applying practical examples, you can elevate your understanding of sugars and their roles in both everyday life and scientific research. Armed with this knowledge, you’re better equipped to make informed choices in the kitchen, the lab, and beyond.