The intricate world of carboxylic acids presents a vital component of organic chemistry, often serving as a cornerstone for more complex organic compounds. Understanding acidity in carboxylic acids is crucial for anyone delving into the field. This guide delves into the nuances of acidity, backed by evidence-based insights and practical applications, providing you with an expert perspective on the subject.
The Essence of Carboxylic Acid Acidity
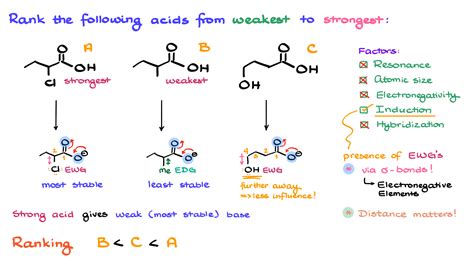
Carboxylic acids exhibit acidity due to the presence of a carboxyl group (-COOH). This functional group endows the molecule with the ability to donate a proton (H+). The acidity of carboxylic acids is primarily governed by their ability to form resonance-stabilized conjugate bases. This resonance stabilization enhances the stability of the anion formed upon the loss of a proton, thus elevating their acidic nature.Key insights box:
Key Insights
- The acidity of carboxylic acids is primarily due to the resonance stabilization of their conjugate bases.
- Electron-withdrawing groups (EWGs) enhance acidity, while electron-donating groups (EDGs) typically reduce it.
- Carboxylic acids often find applications in pharmaceutical synthesis and as starting materials for various organic reactions.
Understanding the Role of Resonance Stabilization
Resonance stabilization is a cornerstone in the study of carboxylic acid acidity. When a carboxylic acid loses a proton, the resulting anion can distribute the negative charge over two oxygen atoms through resonance. This distribution of charge reduces the overall energy of the molecule, leading to greater stability of the anion. For instance, in formic acid (HCOOH), the resonance structure involving delocalization over the two oxygen atoms makes the conjugate base more stable, thereby increasing the acidity of formic acid.Electron Withdrawing and Donating Effects
The presence of substituents around the carboxyl group can significantly influence acidity. Electron-withdrawing groups (EWGs), such as chlorine or nitro groups, increase acidity by drawing electron density away from the carboxyl group, making it easier for the group to donate a proton. Conversely, electron-donating groups (EDGs) like alkyl groups can decrease acidity by pushing electron density towards the carboxyl group, making proton donation less favorable.FAQ section:
What factors influence the acidity of carboxylic acids?
Carboxylic acid acidity is influenced by resonance stabilization, the presence of electron-withdrawing or electron-donating groups, and the overall molecular structure.
How does resonance stabilization contribute to the acidity?
Resonance stabilization enhances the stability of the conjugate base formed when a carboxylic acid donates a proton, thereby increasing its acidity.
In conclusion, the acidity of carboxylic acids hinges on multiple factors, including resonance stabilization and the influence of substituents. A thorough understanding of these elements can significantly aid in predicting and manipulating the behavior of carboxylic acids in various chemical reactions and applications. Whether it’s pharmaceuticals, agrochemicals, or organic synthesis, mastering acidity in carboxylic acids is essential for advanced chemical studies.